Multiple Myeloma and EasyM Resource Centre.

Featured
Minimal Residual Disease as a Primary Endpoint in Multiple Myeloma
Jenna Kerry2025-05-21T11:04:11-04:00May 21st, 2025|Publication|
Opportunities and Challenges for MRD Assessment in the Clinical Management of Multiple Myeloma
Jenna Kerry2025-05-12T11:32:03-04:00May 12th, 2025|Publication|
Publications, Posters & Case Studies
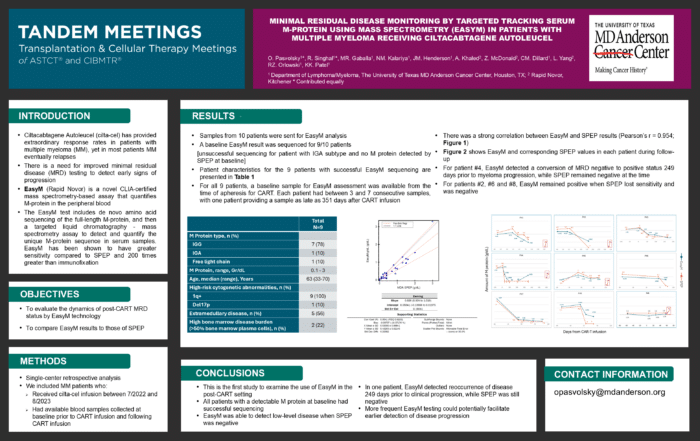
Minimal Residual Disease Monitoring By Targeted Tracking Serum M-Protein Using Mass Spectrometry (EasyM) in Patients with Multiple Myeloma Receiving Ciltacabtagene Autoleucel
Jenna Kerry2026-03-18T15:38:19-04:00March 18th, 2026|Poster|
Sensitive Peripheral Residual Disease (PRD) Detection Using Clonotypic Mass Spectrometry in Patients with Newly Diagnosed Multiple Myeloma (NDMM): The EasyM Approach
Jenna Kerry2026-03-17T17:02:21-04:00March 17th, 2026|Poster|
Clinical Utility of Quantitative, Mass Spectrometry-Based EasyM for MRD Monitoring in Multiple Myeloma: Integration with Established MRD Detection Techniques in a Pan-Canadian Real-World Cohort Study
Jenna Kerry2026-02-24T16:25:21-05:00February 24th, 2026|Poster|
Articles
Extramedullary Disease in Multiple Myeloma
Jenna Kerry2026-04-13T14:50:59-04:00September 3rd, 2025|
Types of Multiple Myleoma
Jenna Kerry2026-04-13T14:47:10-04:00June 16th, 2025|
What is Minimal Residual Disease?
Iain Rogers2025-02-25T22:55:13-05:00February 24th, 2025|
Mass Spectrometry Testing in Multiple Myeloma
Iain Rogers2025-02-24T16:20:23-05:00February 11th, 2025|
M-spike and Multiple Myeloma
Iain Rogers2025-02-24T16:20:40-05:00February 5th, 2025|
Press & News
Rapid Novor Announces Completion of Patient Enrollment in Princess Margaret Cancer Centre Study Evaluating EasyM® in Multiple Myeloma
Jenna Kerry2026-04-07T14:01:46-04:00April 7th, 2026|News|
Rapid Novor Receives Ontario Ministry of Health Provisional License for EasyM® Blood-Based MRD Test for Multiple Myeloma
Jenna Kerry2026-03-10T13:04:52-04:00March 10th, 2026|News|
Monitoring Minimal Residual Disease in Blood with EasyM
Iain Rogers2025-03-14T11:42:06-04:00July 19th, 2024|News|
Rapid Novor Teams Up with PMCC to Evaluate EasyM™ for Multiple Myeloma Minimal Residual Disease Monitoring
Iain Rogers2025-02-24T16:13:03-05:00May 14th, 2024|News|
ASH 2023 | Dr. Ravi Vij – VJ HemeOnc
Iain Rogers2025-03-21T11:58:09-04:00December 14th, 2023|News|
Rapid Novor’s Blood Test for Multiple Myeloma Monitoring is CLIA Certified for Clinical Use in the US
Iain Rogers2026-03-09T16:42:29-04:00March 16th, 2023|News|